Outcomes of Physician-Modified Fenestrated/Branched Endografts for Endovascular Aortic Aneurysm Repair in the Setting of Prior Failed EVAR
Mark Asirwatham1, Varun Konanki1, Spencer J Lucas2, Murray Shames1, Dean J Arnaoutakis1
1University of South Florida, Tampa, FL;2University of South Dakota, Sioux Falls, SD
Objectives: Endovascular treatment of aortic aneurysms involving the renal-mesenteric arteries typically requires a fenestrated/branched endovascular aneurysm repair (F//BEVAR) with a custom-made device (CMD), especially in the setting of prior failed endovascular aneurysm repair (EVAR). However, CMD availability is limited to select centers and thus physician-modified endografts (PMEGs) are an alternative treatment platform. Currently, there is no data on the outcomes of F/BEVAR using PMEG in the setting of failed prior EVAR. The purpose of this study was to evaluate the use of F/BEVAR with PMEG in patients with prior failed EVAR.
Methods: A prospective database of consecutive patients treated at a single-center with F/BEVAR using a PMEG device between March 2021 and August 2022 were retrospectively reviewed. The cohort was stratified by whether there was a failed EVAR present prior to PMEG. Demographics, operative details, postoperative complications were compared between the groups using univariate analysis. One-year survival and freedom from type 1 or 3 endoleak were compared between the groups using Kaplan-Meier method.
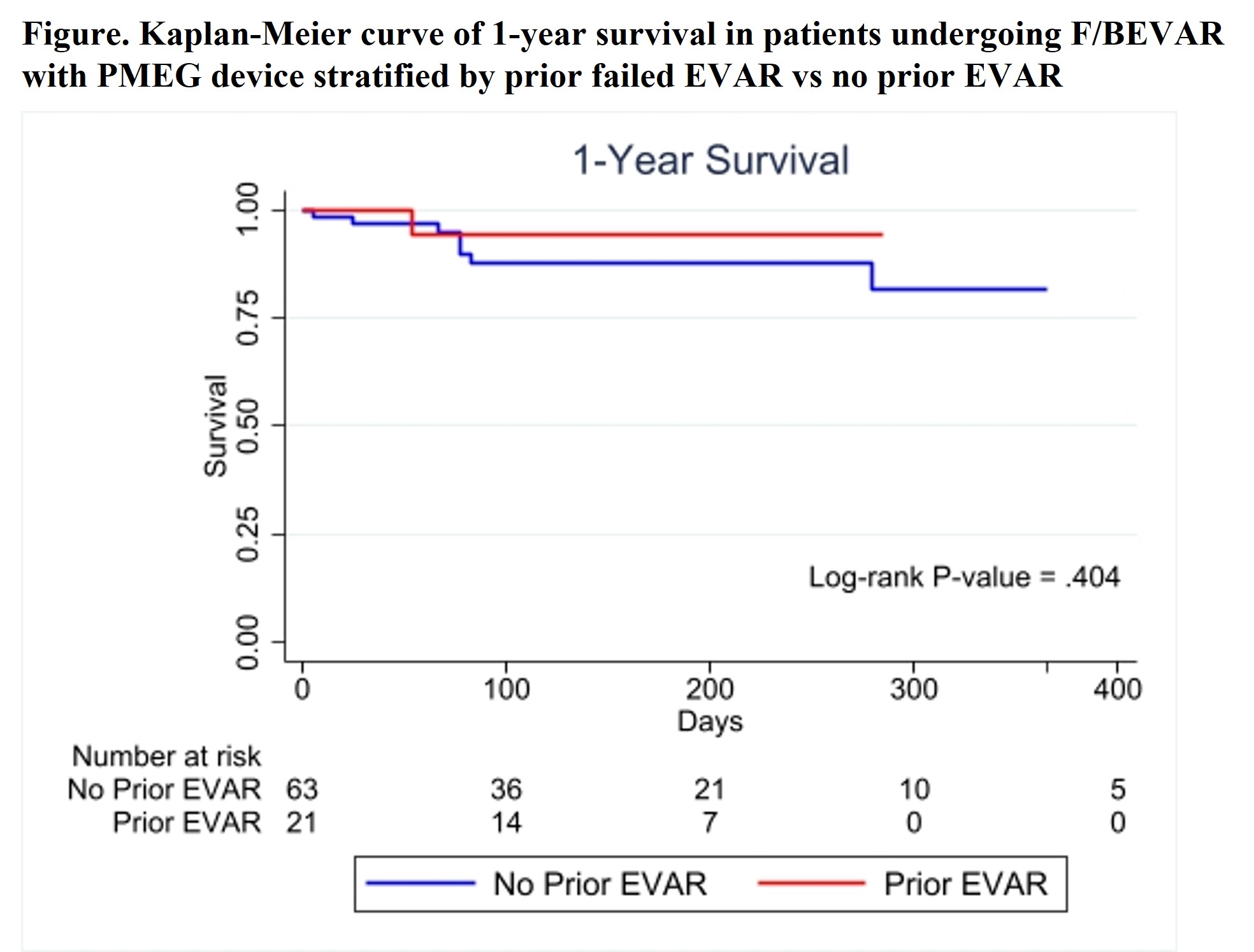
Results: A total of 84 patients underwent F/BEVAR with a PMEG device during the study period, 21 (25%) of which were in the setting of prior failed EVAR. Patients with prior failed EVAR had similar age (75.0�8.3years vs 71.3�9.0; p=.097), male gender (n=19[90%] vs n=46[73%]; p=.135), and comorbid conditions except for higher incidence of moderate-to-severe COPD (n=7[33%] vs n=7[11%]; p=.037) and CAD (n=15[71%] vs n=28[44%]; p=.044). For the entire cohort, aneurysm diameter was 65.4�12.6mm and aneurysm extent was juxtarenal/pararenal in 38% and extent I-V thoracoabdominal in 62%, with no differences between the groups(Table). Twelve (14%) patients presented with symptomatic/ruptured aneurysms. The average number of target arteries incorporated per patient was 3.8. Four different aortic devices were modified with a significantly greater proportion of Terumo TREO devices used in the failed EVAR group (p=.024)(Table). There were no differences in procedure time, radiation dose, or iodinated contrast used between the groups(Table). Overall technical success was 99% and the mean length of stay was 8.3�8.6 days. Rates of postoperative complications (n=6[29%] vs n=14[22%]; p=.564) and 30-day mortality (n=0[0%] vs n=2[3%]; p=1.00) were similar between groups. For the overall cohort, the rates of type 1 or 3 endoleak, branch vessel stenosis/occlusion, and reintervention were 4%, 1%, and 7%, respectively, with no difference between groups. One-year survival (94% vs 82%, p=.404) was similar between groups(Figure).
Conclusions: F/BEVAR using PMEG is a safe and effective treatment for patients with aneurysms involving the renal-mesenteric arteries, even in the setting of prior failed EVAR where additional technical challenges may be present. Additional follow-up is warranted to demonstrate long-term efficacy, but early results are encouraging and similar to those using CMDs.
Table. Operative variables and outcomes of patients who under F/BEVAR with PMEG device
| Variable | Total (n=84) | Failed EVAR (n=21) | No EVAR(n=63) | P-value |
| Aneurysm Extent | .284 | |||
| Juxtarenal | 6 (7) | 1 (5) | 5 (8) | |
| Pararenal | 26 (31) | 7 (33) | 19 (30) | |
| Extent I TAAA | 5 (6) | 0 (0) | 5 (8) | |
| Extent II TAAA | 6 (7) | 1 (5) | 5 (8) | |
| Extent III TAAA | 13 (15) | 1 (5) | 12 (19) | |
| Extent IV TAAA | 26 (31) | 10 (48) | 16 (25) | |
| Extent V TAAA | 2 (2) | 1 (5) | 1 (2) | |
| Maximum aneurysm diameter, mm | 65.4 (12.6) | 67.0 (10.4) | 64.8 (13.3) | .481 |
| Number target arteries incorporated/patient | 3.8 (0.6) | 3.7 (0.7) | 3.8 (0.6) | .37 |
| Aortic device modified | .024 | |||
| Cook Alpha | 17 (20) | 2 (10) | 15 (24) | |
| Cook TX2 | 17 (20) | 1 (5) | 16 (25) | |
| Cook ZFen | 1 (1) | 0 (0) | 1 (2) | |
| Terumo Aortic TREO | 49 (58) | 18 (86) | 31 (49) | |
| Dose area product, Gy x cm2 | 439 (239) | 361 (166) | 466 (255) | .081 |
| Cumulative air kerma, mGy | 3,052 (1,613) | 2,834 (1,657) | 3,128 (1,604) | .476 |
| Iodinated contrast use, mL | 80 (27) | 73 (22) | 83 (28) | .155 |
| Fluoroscopy time, minutes | 78 (37) | 80 (39) | 77 (37) | .754 |
| Procedure time, minutes | 243 (108) | 255 (122) | 239 (103) | .564 |
| Technical Success | 83 (99) | 21 (100) | 62 (98) | 1.00 |
| Length of stay, days | 8.3 (8.6) | 7.4 (4.7) | 8.6 (9.5) | .590 |
| Any postoperative complication | 20 (24) | 6 (29) | 14 (22) | .564 |
| Access site complication | 3 (4) | 1 (5) | 2 (3) | .250 |
| Myocardial infarction | 1 (1) | 0 (0) | 1 (2) | 1.00 |
| Respiratory complication | 8 (10) | 3 (14) | 5 (8) | .406 |
| Acute renal failure | 8 (10) | 3 (14) | 5 (8) | .406 |
| Permanent spinal cord ischemia | 3 (4) | 1 (5) | 2 (3) | 1.00 |
| 30-day mortality | 2 (2) | 0 (0) | 2 (3) | 1.00 |
| Reintervention | 6 (7) | 2 (10) | 4 (6) | .637 |
| Any endoleak | 21 (29) | 4 (20) | 17 (33) | .390 |
| Type I or III endoleak | 3 (4) | 0 (0) | 3 (6) | .555 |
| Branch vessel stenosis/occlusion | 1 (1) | 0 (0) | 1 (2) | 1.00 |
Back to 2023 Display Posters


